 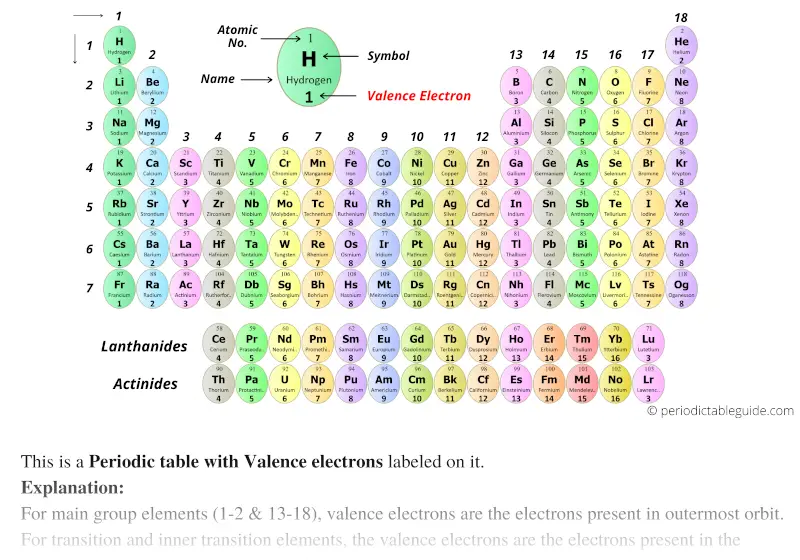
However, as n increases, note this becomes less noticeable and negligible. Oxygen meanwhile has 4 valence electrons, and it would be more stable with only half a subshell so it takes less energy to remove the electron, which we say is due to electron-electron repulsion forces. Each hydrogen atom (group 1) has one valence electron, carbon (group 14) has 4 valence electrons, and oxygen (group 16) has 6 valence electrons, for a total of (2)(1) + 4 + 6 12 valence electrons. However one exception is Oxygen having a lower ionization energy than nitrogen-this is because of subshell occupancy! Nitrogen has 3 electrons int he 2p valence subshell, which is halfway! This would mean it has 3 electrons of the same spin in the subshell. (Recall that the number of valence electrons is indicated by the position of the element in the periodic table.). Using the Periodic Table The last digit of the Group (column) an element is in is its number of valence electrons. The most energy to remove is going to be helium! Therefore, as you move down a period (row) the ionization energy increases. 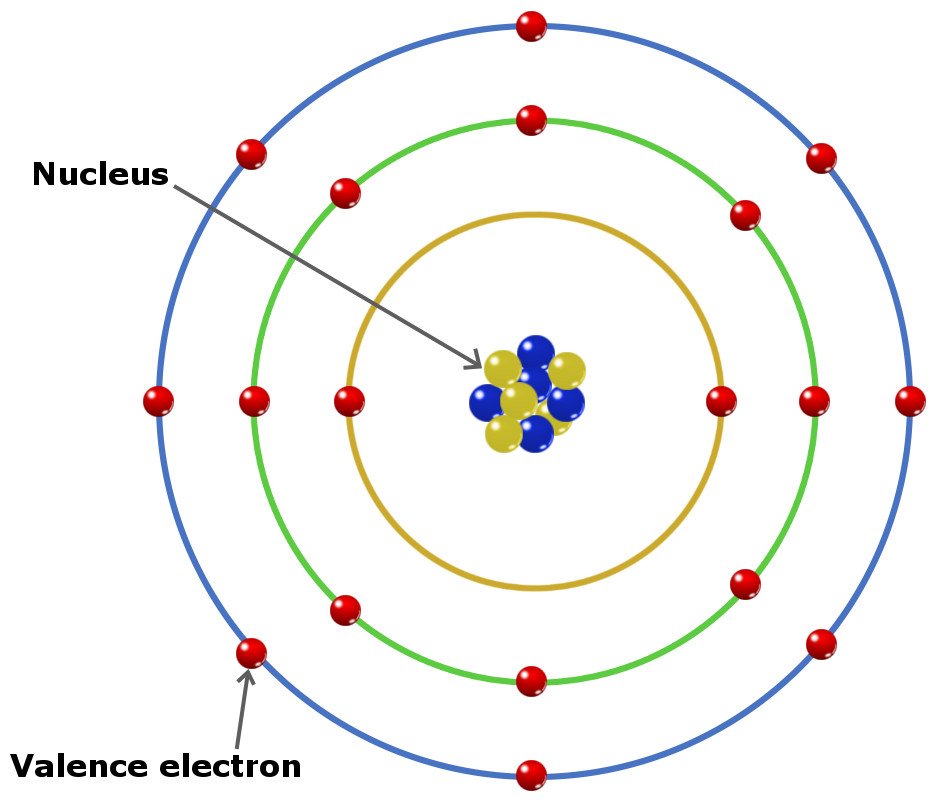
Meanwhile, all the noble gasses are completely filled in subshells so they REALLY dont wanna give away an electron, so it takes even more energy to remove. For atoms like flourine, it is SO close to getting that 6th electron that it takes a lot fo energy to remove an electrona nd take it away from its goal. The number of bonds that an atom can form can often be predicted from the number of electrons needed to reach an octet (eight valence electrons) this is especially true of the nonmetals of the second period of the periodic table (C, N, O, and F). Subshell occupancy also plays a big role. Elements in the same group have the same number of valence electrons and therefore. So, then as shell number increases, the less the electron feels attached to the nucleus of the atom, so the easier it will be for it to be removed-so ionization energy decreases down groups (columns) on the periodic table since the n increases as you go "down". Valence electrons determine an elements chemical properties and reactivity. Some things that impact how much the electron is 'happy' are: shell number (n) as the further you are from the nucleus the less it is pulled to it (like how you feel the heat of a campfire the closer you are to it and the less people in front of you), subshell occupancy. How much energy it takes to remove the electron is directly tied to how much the electron is 'attached' to staying with its atom. The ionization energy is going to be the amount of energy it takes to remove an electron from the valence shell of an atom. Not every atom has the same number of valence electrons, it depends on the location of the element on the periodic table. Group number Number of valence electrons. Hi! I have a question - I understand but Im still a bit confused so can someone explain to me how does the trend in ionization energy vary across the periodic table, and what factors contribute to this trend? Thanks! Thus, members of a family in the periodic table share some properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
